Keep you informed
Welcome to the Galenicum Axium news section. In this site, we will inform you about the latest updates of the company together with the most relevant product updates.
In Galenicum Axium, we are committed to our customers and partners, and we want to keep you informed with all the latest achievements of the company and the projects we are working on.
Galenicum NEWS

PharmaVenue 2022 Marbella
Galenicum will be attending PharmaVenue 2022 in Marbella December 1st and 2nd.
We have already booked most of our slots. However, if you still do not have an appointment but would like to discuss potential business opportunities, do not hesitate to contact us.
VISIT US AT CPhI FRANKFURT 2022
CPhI 2022 Frankfurt
Galenicum will be exhibiting at the CPhI Worldwide 2022 in Frankfurt from November 1st to 3rd.
We have already booked slots with most of our partners. However, if you still do not have an appointment but would like to discuss potential business opportunities, come and meet us at Hall 4.1 (FDF), Stand H20. We are very excited to meet you in person after such a long time!

New York - DCAT WEEK

Galenicum visits New York during DCAT Week
After Galenicum's attendance at the CPhI in Milano the last month of November, it is time for new pharma events. We are visiting DCAT Week in New York City from 21st until 24th of March, 2022.
For one week, Galenicum will be in New York in order to meet partners, and to potentially find new ones. Are you willing to meet any of our representatives in the event? Do not hesitate to contact us at hello@galenicum.com
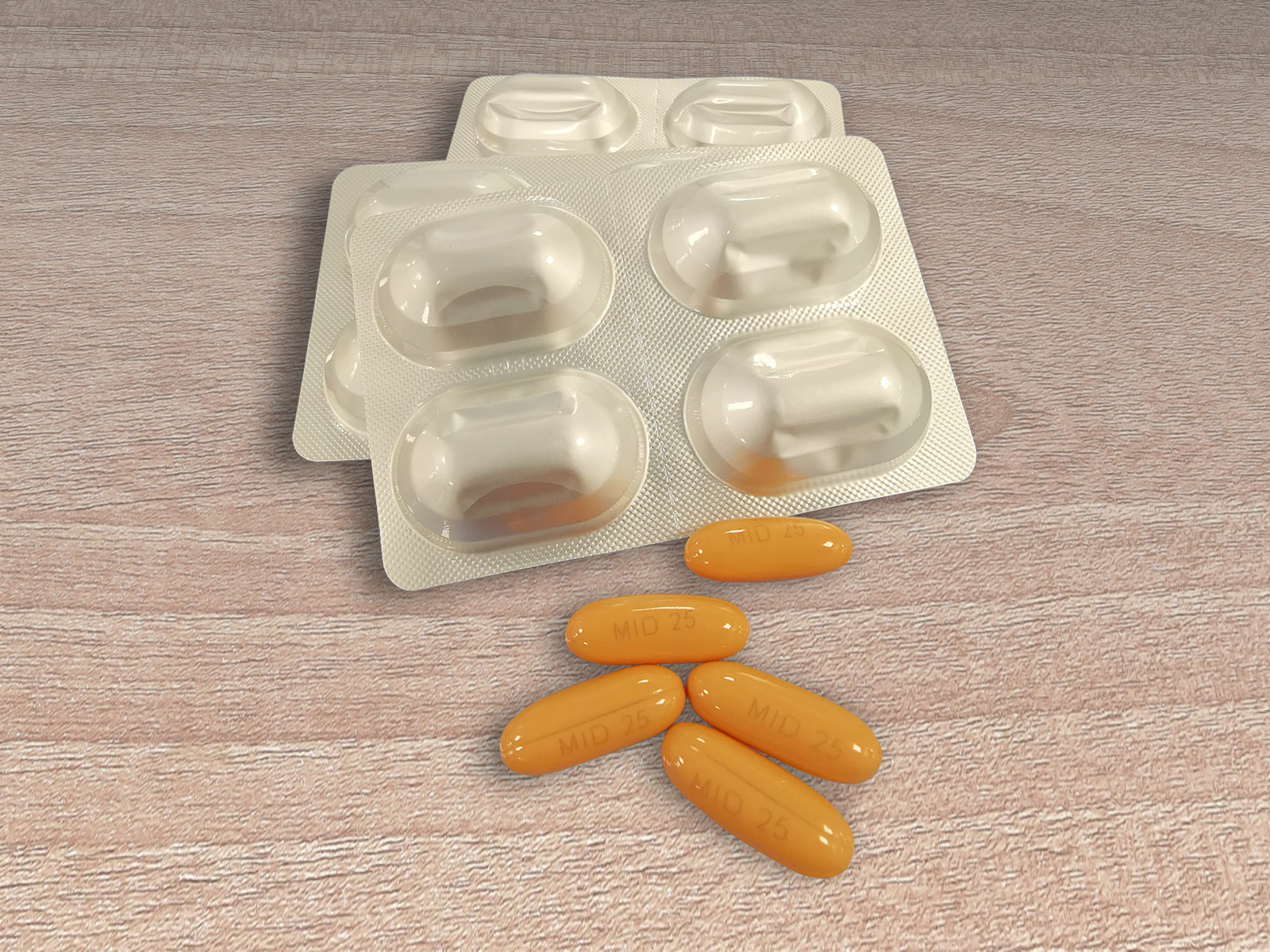
Midostaurin NCE-1
Galenicum partner files Midostaurin in the US
In 2021, Galenicum Axium accomplished an important milestone with the submission of Midostaurin soft gel capsules 25mg Dossier to the US FDA on the NCE-1 date.
Midostaurin is an oral prescription medicine used to treat adults with a certain type of Acute Myeloid Leukemia (AML), and whose innovator is RYDAPT® from Novartis.
Living up to our mission of improving the quality and affordability of pharmaceutical products worldwide, Midostaurin has been a great addition to our broad portfolio of products. Are you interested? Please, contact us or continue reading.
Visit us at CPhI Milano 2021

CPhI 2021 Milano
Galenicum will be exhibiting at the CPhI Worldwide 2021 in Milan from November 9th to 11th.
We have already booked slots with most of our partners. However, if you still do not have an appointment but would like to discuss potential business opportunities, come and meet us at Hall 1 (FDF), Stand F31. We are very excited to meet you in person after such a long time!
